Get Smarter and Efficient MLR Reviews with ERMA Evaluate™
ERMA Evaluate uses purpose-built AI to identify claims, assign references, and streamline MLR workflows — helping life sciences teams move faster while maintaining regulatory confidence. Instead of maintaining static claims libraries, ERMA analyzes promotional content directly, ensuring every asset enters review with claims identified, references assigned, and labeling alignment verified.
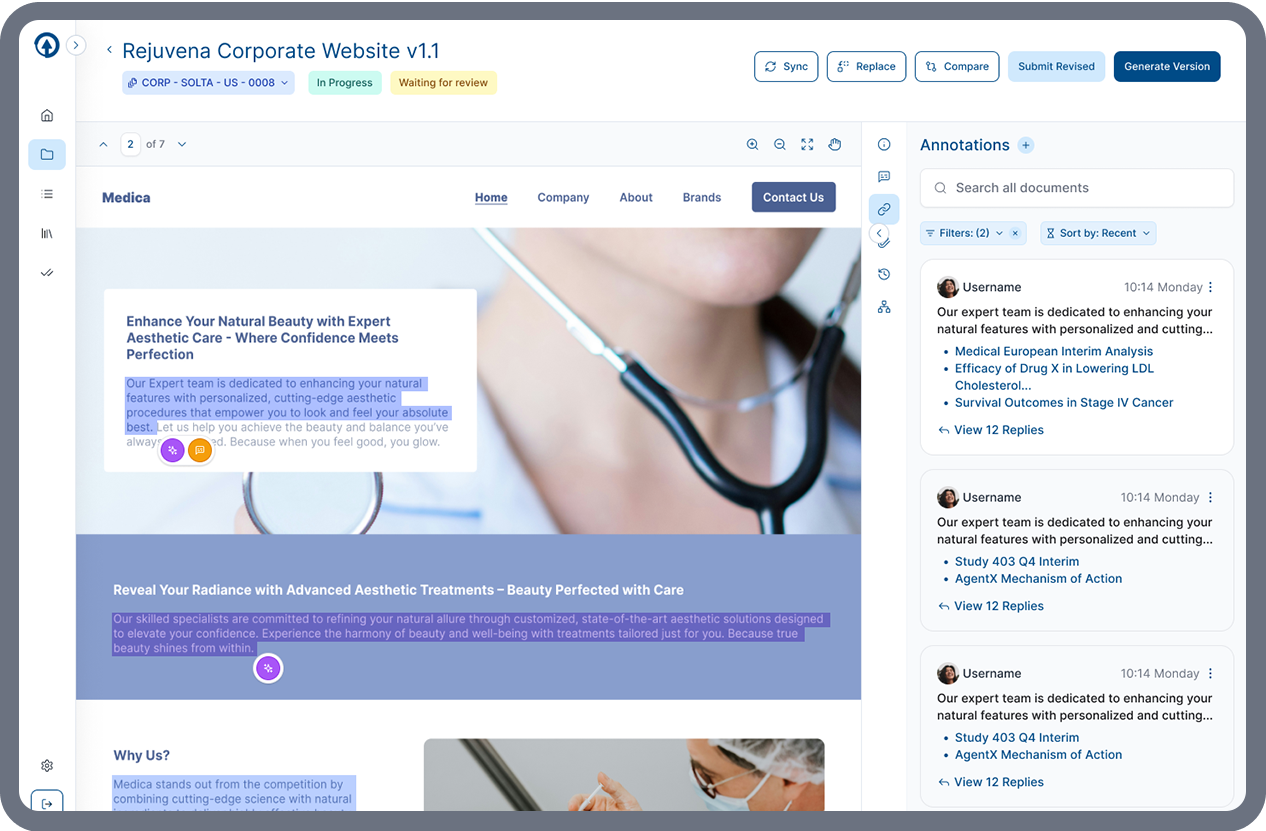
AI-Powered Claim Identification and Reference Assignment
ERMA Evaluate automatically detects scientific and promotional claims directly within marketing materials — eliminating the need to manually create and maintain claims databases. By identifying claims dynamically within the content, ERMA ensures reviewers can focus on evaluating messaging rather than managing claims infrastructure.
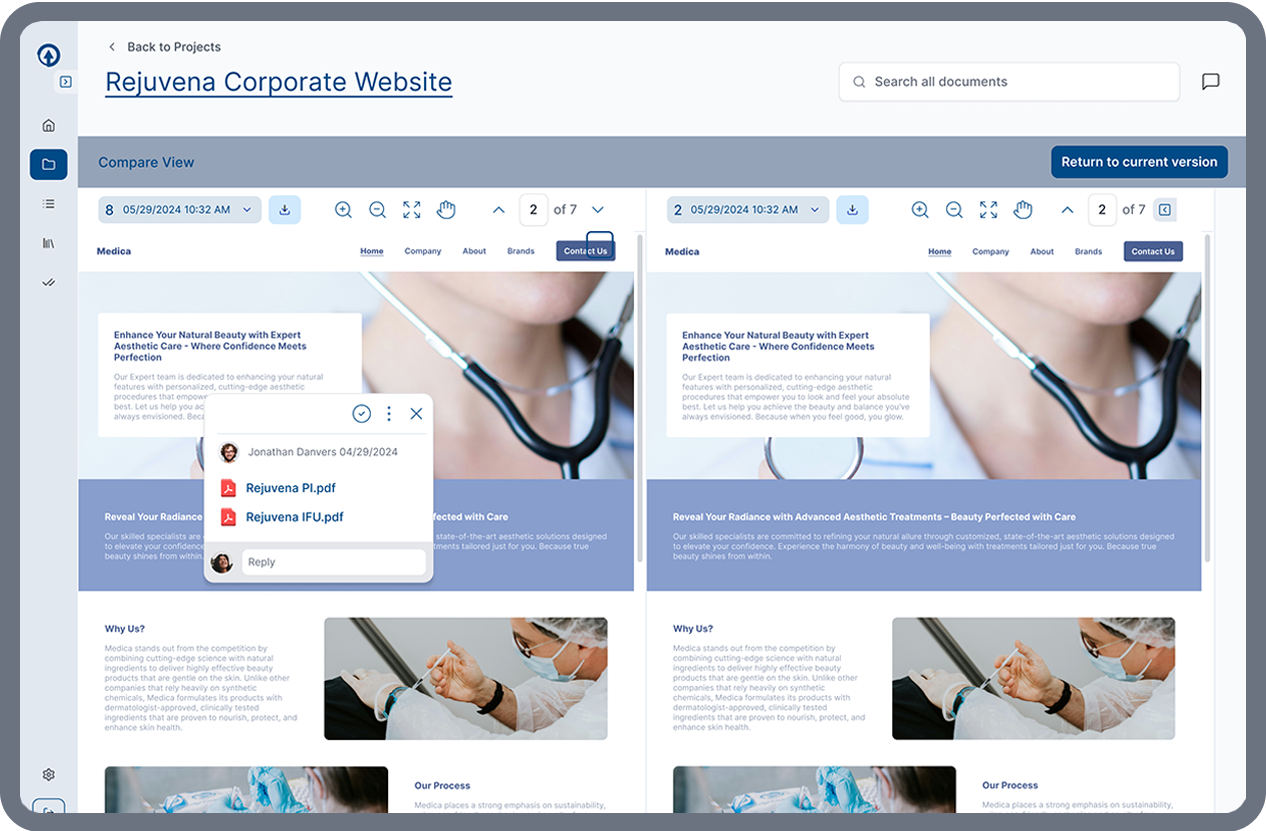
Side-by-Side Content Comparison
ERMA Evaluate allows reviewers to compare versions of promotional materials instantly. Side-by-side comparisons highlight content changes, making it easier for reviewers to evaluate updates and approve revisions without unnecessary back-and-forth.
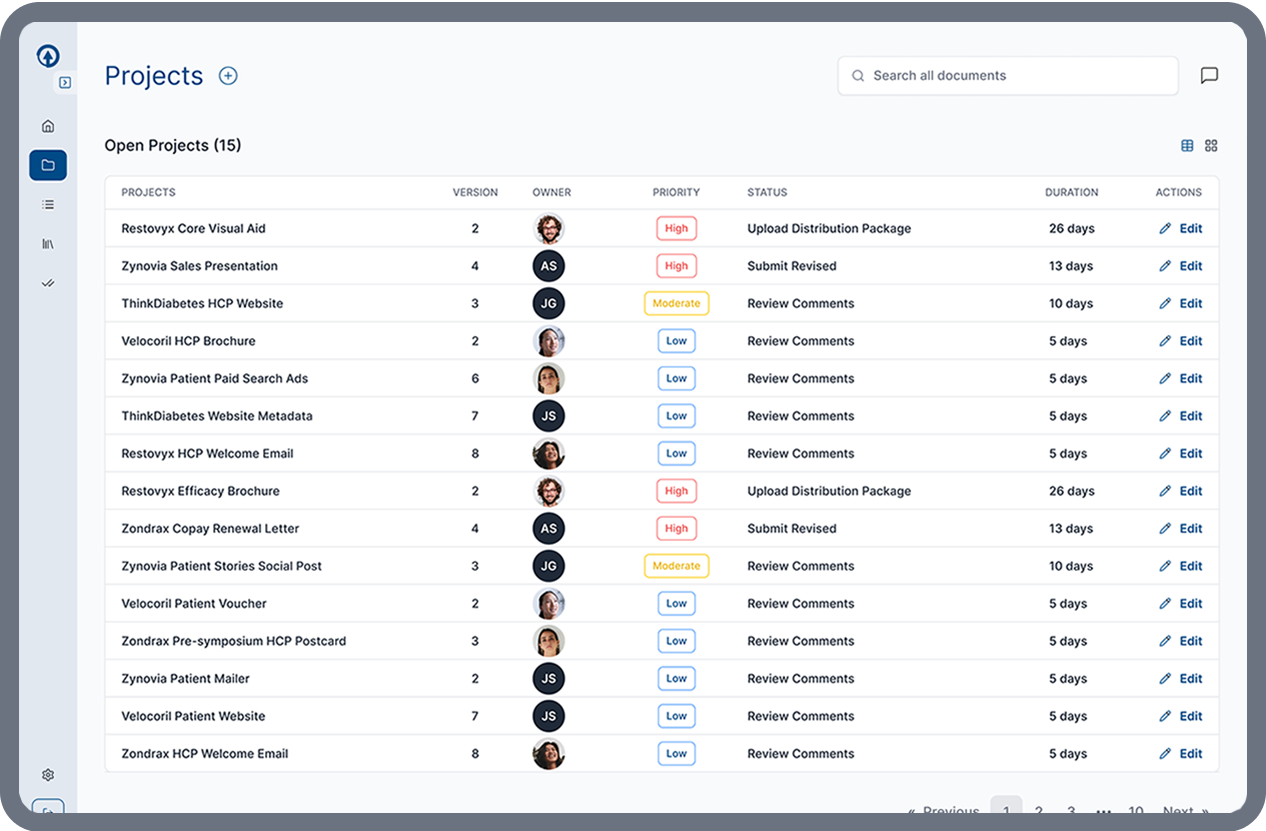
Flexible MLR Workflows
Every organization structures promotional review differently. ERMA Evaluate provides configurable workflows that adapt to your internal processes rather than forcing teams into rigid review structures.
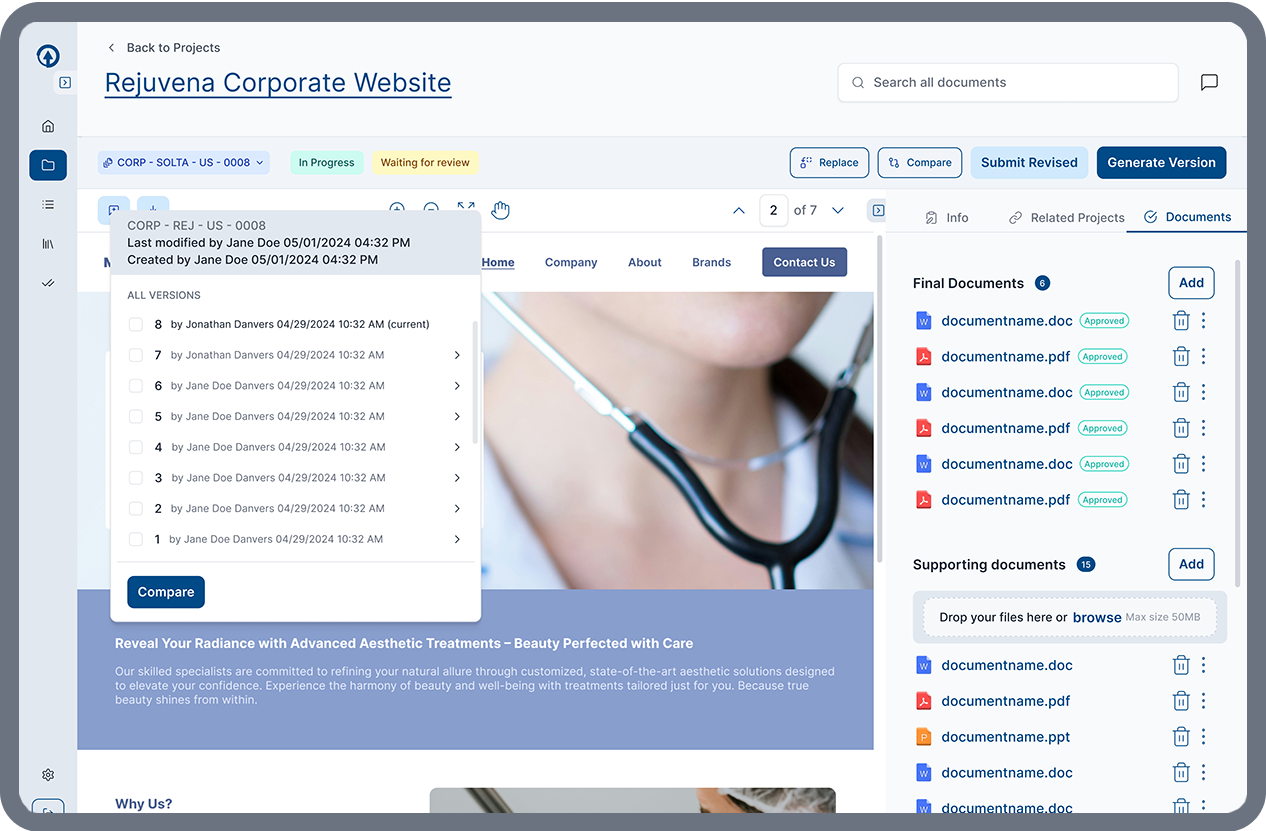
FDA-Ready Documentation
ERMA Evaluate helps teams prepare approved materials for regulatory submission by organizing required documentation and metadata for streamlined eCTD 2253 preparation. This helps teams move from approval to submission faster while maintaining regulatory traceability.
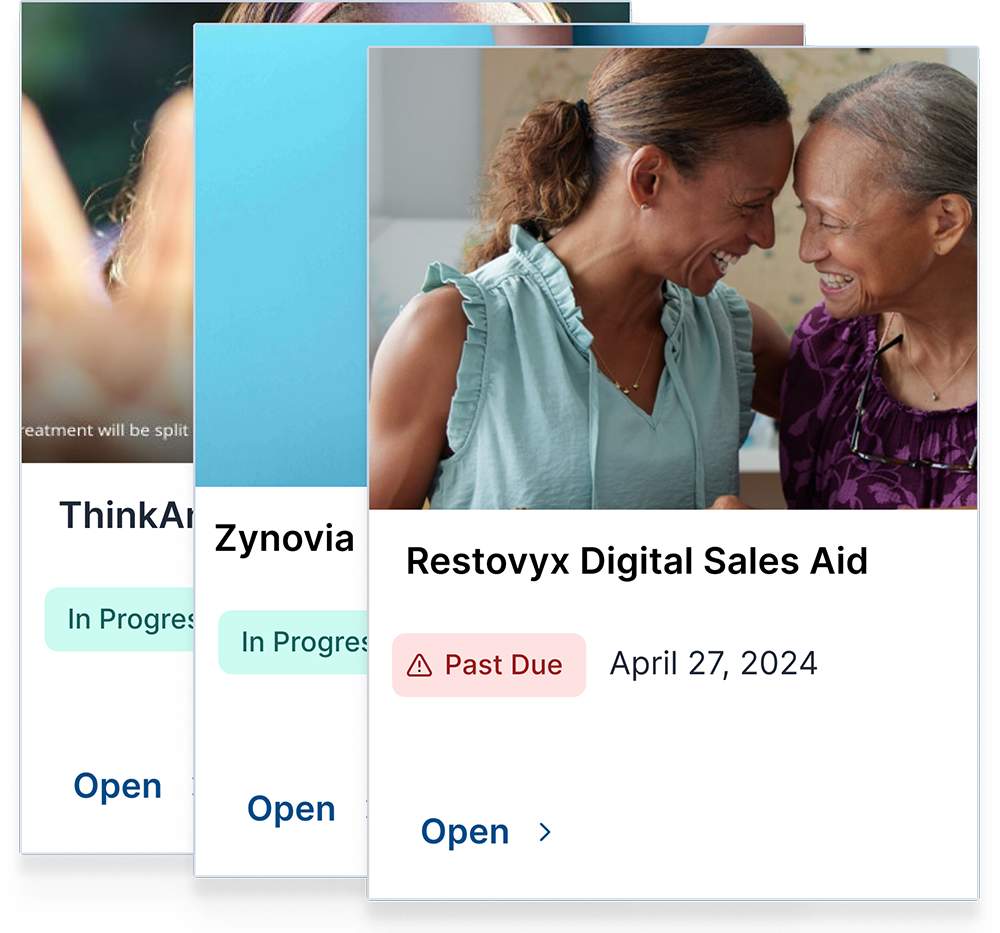
Expiration Tracking & Re-Approval Management
ERMA Evaluate continuously monitors approved materials and proactively alerts teams when references, labeling, or content require re-approval. This ensures organizations maintain compliant promotional materials without relying on manual tracking.